|
The effect of salt formation on structure and charge distribution in imines. Thermochromism and photochromism of aryl-substituted acyclic azines. A dynamic NMR study of Z, E‐isomerization in solutions of indolyl‐substituted α‐nitroacrylates. Amide-oximes: determination des configurations et etude du mecanisme de l’isomerisation Z– E. Thermal isomerization about double bonds. Effects of bond-angle inversion on the statistical properties of poly(dimethylsiloxane). A molecular orbital exploration of structure, stereochemistry, and reaction dynamics. 
Description of real molecules referenced to idealized polygons or polyhedra in geometric reaction path form. Detection of polytopal isomers in the solution state. Structure and stereochemical nonrigidity of six-coordinate complexes. Topological representation of stereoisomerism. Topological analysis of polytopal rearrangements. Phosphororganische verbindungen optisch aktive tertiäre Phosphine aus optisch aktiven quartären Phosphoniumsalzen. In Molekulare Asymmetrie 803–824 (Franz-Deutike, Leipzig, 1933). The resolution of γ-6:6′-dinitro- and 4:6:4′:6′-tetranitro-diphenic acids into optically active components. The molecular configurations of polynuclear aromatic compounds. The configuration of diphenyl and its derivatives. The possibility of a new instance of optical activity without an asymmetric carbon atom. Prostereoisomerism and the concept of product stereoselectivity in biochemistry and xenobiotic metabolism. The concept of substrate stereoselectivity in biochemistry and xenobiotic metabolism. The conformation factor in molecular pharmacology. Stereoselectivity in molecular and clinical pharmacology. Isomerisms about single bonds and in cyclic systems. Other stereogenic elements: axes of chirality, planes of chirality, helicity, and ( E, Z)-diastereoisomerism. Stereoisomerism resulting from one or several stereogenic centers. Symmetry elements and operations, classification of stereoisomers. Organic stereochemistry: guiding principles and bio-medicinal relevance. IUPAC Compendium of Chemical Terminology - The Gold Book 2nd edn (Blackwell, Oxford, 1997). Based on an extended polytope formalism of molecular structure and stereoisomerization, BAI-driven akamptisomerization is shown to be the final fundamental type of conformational isomerization. We also introduce the necessary BAI stereodescriptors parvo and amplo. 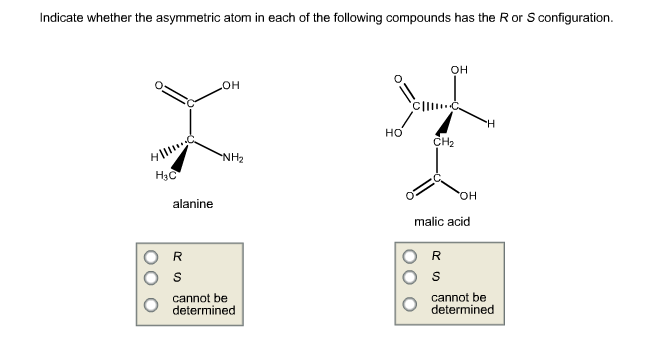
Feasible interconversion processes between conceivable synthesis products and reaction intermediates were mapped out by density functional theory calculations, identifying bond-angle inversion (BAI) at a singly bonded atom as the reaction mechanism. 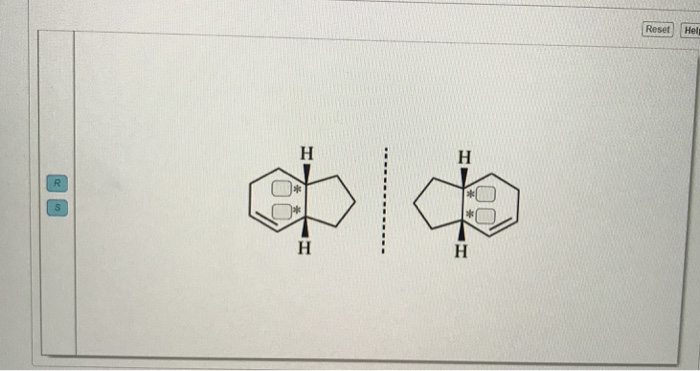
They undergo thermal diastereomeric interconversion over a barrier of 104 ± 2 kJ mol −1, which we term ‘akamptisomerization’. 
These comprise two pairs of enantiomers that manifest structural relationships not describable within existing IUPAC nomenclature and terminology. Here we describe a previously unclassified fundamental form of conformational isomerism through four resolved stereoisomers of a transoid (BF)O(BF)-quinoxalinoporphyrin. Isomerism is a fundamental chemical concept, reflecting the fact that the arrangement of atoms in a molecular entity has a profound influence on its chemical and physical properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
